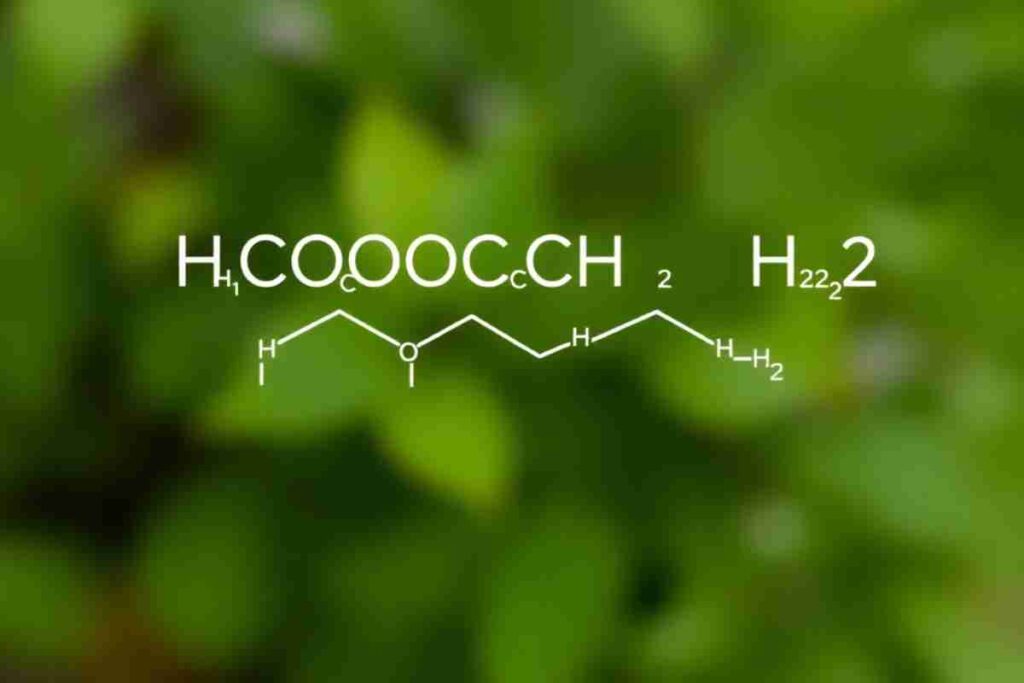
HCOOCH CH₂ H₂O is an organic compound with a vital role in chemical synthesis, industrial production, and pharmaceutical applications. It features ester and hydroxyl functional groups, making it reactive in multiple environments. Chemists use it in organic reactions, solvents, and drug synthesis, making it essential in laboratories and industries.
- Chemical Components and Their Roles
- HCOO (Formate group): Acts as an electron donor in reactions.
- CH₂ (Methylene group): Provides structural integrity and connects functional groups.
- H₂O (Water molecule): May indicate hydration or reaction byproducts.
- Applications in Various Fields
HCOOCH CH₂ H₂O is widely used in organic synthesis, polymer production, and medicine. Researchers explore its potential in green chemistry, drug formulations, and biofuels, making it a compound of interest for future applications.
Understanding the Chemical Structure of HCOOCH CH₂ H₂O
 Molecular Breakdown
Molecular Breakdown
HCOOCH CH₂ H₂O consists of formate (HCOO), a methylene group (CH₂), and water (H₂O). These components influence the compound’s reactivity, solubility, and bonding properties.- Functional Groups Present
- Ester (-COO-): Enhances solubility and reactivity.
- Hydroxyl (-OH): May affect polarity and hydrogen bonding.
- Alkyl (-CH₂-): Influences volatility and chemical stability.
- Structural Representation and Molecular Bonding
| Element | Atomic Symbol | Role in Molecule |
| Carbon | C | Backbone of the molecule |
| Hydrogen | H | Involved in bonding and reactivity |
| Oxygen | O | Forms ester and hydroxyl groups |
Formation and Synthesis of HCOOCH CH₂ H₂O
- Chemical Reactions Producing HCOOCH CH₂ H₂O
HCOOCH CH₂ H₂O forms through esterification reactions involving formic acid and an alcohol. Catalysts such as sulfuric acid or enzymes help drive the reaction forward.
Reaction Equation:
HCOOH+CH2OH→HCOOCHCH2H2OHCOOH + CH₂OH → HCOOCH CH₂ H₂OHCOOH+CH2OH→HCOOCHCH2H2O - Conditions for Synthesis
- Requires acidic conditions for esterification.
- Optimum temperature of 60-80°C.
- Reaction time of 2-6 hours depending on the catalyst.
- Industrial and Laboratory Methods
- Industrial: Large-scale production using acid catalysts.
- Laboratory: Small-scale synthesis using drying agents to remove water byproducts.
Physical and Chemical Properties of HCOOCH CH₂ H₂O

- Physical Properties
- State: Liquid
- Boiling Point: ~120-150°C
- Solubility: Miscible with alcohols and slightly soluble in water
- Chemical Behavior in Different Environments
- In Acidic Solutions: Undergoes hydrolysis, breaking down into formic acid and alcohol.
- In Basic Solutions: Forms carboxylate salts, increasing solubility.
- Stability and Reactivity
- Stable under normal storage conditions.
- Reacts with strong acids and bases, requiring careful handling.
HCOOCH CH₂ H₂O in Organic Chemistry
- Role in Organic Synthesis
HCOOCH CH₂ H₂O serves as a precursor in esterification and condensation reactions. It helps in forming esters, ethers, and polymers used in industries. - Common Derivatives and Related Compounds
- Methyl formate (HCOOCH₃): Used in perfumes and solvents.
- Ethyl formate (HCOOCH₂CH₃): Found in flavoring agents.
- Importance in Chemical Industry and Pharmaceuticals
- Acts as a solvent in drug formulations.
- Used in synthetic flavorings and polymer resins.
Uses and Applications of HCOOCH CH₂ H₂O
- Industrial Applications
- Used in solvent manufacturing for coatings and adhesives.
- Helps in organic synthesis to create high-value chemicals.
- Biochemical and Pharmaceutical Role
- Functions as an intermediate in drug synthesis.
- Used in ester-based medications.
- Future Applications and Research Areas
- Green chemistry: Sustainable processes for eco-friendly solvents.
- Biodegradable materials: Potential use in eco-friendly polymers.
Reactions Involving HCOOCH CH₂ H₂O

- Common Reactions
- Hydrolysis: Breakdown into formic acid and alcohol.
- Esterification: Formation of new ester derivatives.
- Interaction with Acids, Bases, and Catalysts
- Strong acids catalyze hydrolysis and esterification.
- Bases lead to carboxylate salt formation.
- Reaction Mechanisms and Examples
| Reaction Type | Equation |
| Hydrolysis | HCOOCH CH₂ H₂O + H₂O → HCOOH + CH₂OH |
| Esterification | HCOOH + CH₂OH → HCOOCH CH₂ H₂O |
Toxicity and Safety Considerations of HCOOCH CH₂ H₂O
- Potential Health Hazards
- Exposure may cause skin and eye irritation.
- Inhalation of vapors can lead to respiratory discomfort.
- Prolonged exposure may affect the nervous system and liver function.
- Proper Handling and Storage Guidelines
- Store in airtight containers away from heat and direct sunlight.
- Use protective gloves and goggles when handling.
- Ensure proper ventilation to prevent inhalation risks.
- Environmental Impact and Safety Measures
- Biodegradable in water but may cause mild aquatic toxicity.
- Dispose of in accordance with hazardous waste regulations.
- Use spill containment methods to prevent soil and water contamination.
HCOOCH CH₂ H₂O in Industrial Manufacturing
- Role in Chemical Production and Industrial Processes
- Used as a reactant in esterification for various chemical formulations.
- Essential in the synthesis of polymers, resins, and solvents.
- Large-Scale Production Techniques
- Produced through catalytic esterification in continuous reactors.
- Requires temperature-controlled distillation to ensure purity.
- Economic and Commercial Importance
- High demand in pharmaceuticals, cosmetics, and synthetic materials.
- Cost-effective due to simple raw materials and scalable synthesis.
HCOOCH CH₂ H₂O in Biological Systems
- Possible Biological Roles and Metabolic Pathways
- May serve as an intermediate in metabolic ester reactions.
- Can be hydrolyzed by enzymes into formic acid and alcohol derivatives.
- Effects on Human Health and the Environment
- Low acute toxicity, but long-term exposure may affect liver metabolism.
- Decomposes into non-toxic byproducts in biological environments.
- Research on Biological Interactions
- Studies focus on its biodegradation potential.
- Investigated for pharmaceutical applications as a solvent or reactant.
Comparison of HCOOCH CH₂ H₂O with Similar Compounds
- Structural and Functional Similarities
- Shares ester properties with methyl formate and ethyl formate.
- Similar reactivity to acetates and propionates.
- Differences in Reactivity, Stability, and Uses
| Compound | Reactivity | Stability | Common Uses |
| HCOOCH CH₂ H₂O | Moderate | High | Solvents, pharmaceuticals |
| Methyl Formate (HCOOCH₃) | High | Moderate | Flavoring agents, adhesives |
| Ethyl Formate (HCOOCH₂CH₃) | High | Low | Perfumes, coatings |
- Comparison in Industrial and Laboratory Applications
- HCOOCH CH₂ H₂O has better thermal stability than ethyl formate.
- Less volatile than methyl formate, making it safer for storage.
Conclusion on HCOOCH CH₂ H₂O
HCOOCH CH₂ H₂O is a versatile ester with significant applications in chemistry, industry, and biological systems. Its chemical stability, moderate reactivity, and solubility make it valuable in organic synthesis, pharmaceuticals, and industrial manufacturing. Despite its mild toxicity, proper handling and storage ensure safe usage.
Future research aims to develop eco-friendly synthesis methods, enhance biodegradability, and explore new pharmaceutical applications. As the demand for sustainable and efficient chemicals grows, HCOOCH CH₂ H₂O will continue to play a crucial role in scientific advancements. Its potential in biodegradable polymers, medical research, and industrial applications highlights its long-term importance in the field of chemistry.